Kalmomin Turanci masu kama da juna: ci37500;ciazoiccouplingcomponent1;cideveloper5;ChemicalbookDeveloperA;DeveloperAMS;DeveloperBN;developer;developerams
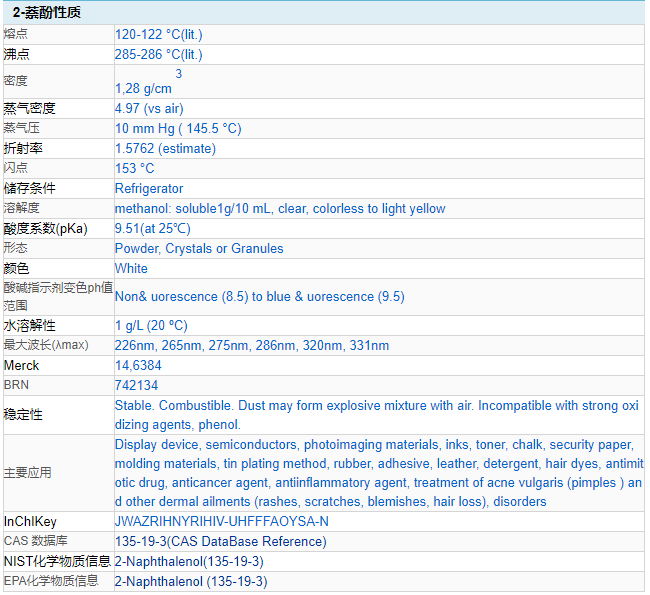
Lambar CAS: 135-19-3
Tsarin kwayoyin halitta: C10H8O
Nauyin kwayoyin halitta: 144.17
Lambar EINECS: 205-182-7
Rukuni masu alaƙa:
Matsakaici; Ma'adanai Masu Ƙamshi; Launi na Tsoffin Ma'adanai; Mai Haɓakawa; Haruffa; Ƙananan Kwayoyin Halitta Masu Aiki; Sinadaran Halittu da Maganin Halittu; Toshewar Gine-gine; Barasa; MICROCIDIN; Alamu; Mai Haske; Naphthalene; Mai Launi Mai Tsami; Ƙamshi; Mai Launi Mai Tsami; Dabbobi Masu Tsami; BioxyLabs; Mai Haske; Naman Halittu; Bigenology; Sinadaran Halitta; Barbashi da Tabo; Masu Ƙarfin Probes da Alamomi;
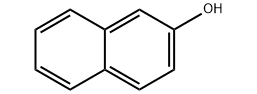
Hanyar amfani da naphthol 2 da kuma hadawa
| Matsakaicin izinin amfani da ƙarin abinci da matsakaicin ƙa'idodin ragowar da aka yarda da su | Sunan ƙari | Sunan abincin da ke ba da damar amfani da wannan ƙarin | Aikin ƙari | Matsakaicin amfani da aka yarda da shi (g/kg) | Matsakaicin ragowar da aka yarda da shi (g/kg) |
| Acetonitrile | 'Ya'yan itatuwa sabo da aka yi wa magani a saman ('ya'yan itacen citrus kawai) | abin hana tarawa | 0.1 | Adadin da ya rage ≤70mg/kg |
Kayayyakin sinadarai:Farin ƙuraje masu sheƙi ko farin foda. Ba ya narkewa a cikin ruwa, yana narkewa a cikin maganin ethanol, ether, chloroform, glycerin da alkali.
Amfani:
1. Ana amfani da shi wajen shirya sinadarin Tobias acid, J acid, 2,3 acid da azo dyes, kuma kayan aiki ne na roba don hana tsufa, magungunan sarrafa ma'adanai, magungunan kashe ƙwayoyin cuta, magungunan kashe ƙwayoyin cuta, magungunan kiyayewa, da sauransu.
2. A matsayin abin kiyayewa, ƙasata ta tanadar da cewa za a iya amfani da shi don kiyaye citrus, matsakaicin adadin amfani shine 0.1g/kg, kuma ragowar adadin bai wuce 70mg/kg ba.
3. Ethylnaphthol, wanda aka fi sani da β-naphthol da 2-naphthol, wani sinadari ne na naphthoxyacetic acid mai daidaita girman shuka.
4. Ana amfani da shi azaman abin kiyaye abinci. A cewar ƙasarmu, ana iya amfani da shi don kiyaye citrus. Matsakaicin adadin amfani shine 0.1g/kg, kuma ragowar adadin bai wuce 70mg/kg ba.
5. Ana amfani da shi azaman reagent na nazari, ethylene, mai shan carbon monoxide da kuma alamar haske
6. Muhimman kayan albarkatun ƙasa na halitta da kuma tsaka-tsakin rini, waɗanda ake amfani da su wajen ƙera Toubic acid, butyric acid, β-naphthol-3-carboxylic acid, kuma ana amfani da su wajen ƙera maganin hana tsufa D, maganin hana tsufa DNP da sauran magungunan hana tsufa, pigments na halitta da magungunan kashe ƙwayoyin cuta. Jira.
7. Ana amfani da shi wajen shirya sinadarin Tobias acid, J acid, 2.3-acid, kuma ana amfani da shi wajen shirya sinadarin D, sinadarin DNP da magungunan da ke hana tsufa, da kuma sinadaran da ke hana tsufa da kuma magungunan fungi.
8. Tabbatar da bromine, chlorine, chlorate, niobium, jan ƙarfe, nitrite da potassium. ...
9. Tabbatar da bromine, chlorine, chlorate, niobium, jan ƙarfe, nitrite da potassium; tantance carbon monoxide, jan ƙarfe, nitrite da potassium; tantance inganci na allyl alcohol, methanol, chloroform, da sauransu; transferase phenolsulfon substrate ma'aunin haske; mai sha ethylene; mai nuna fluorescent; mai nuna tushen acid; tsaka-tsaki na rini.
Hanyar samarwa:
1. An yi shi ne da naphthalene ta hanyar sulfonation da kuma haɗakar alkali. Hanyar haɗakar alkali da aka yi da sulfonated alkali hanya ce da ake amfani da ita sosai a gida da kuma ƙasashen waje, amma tana da tsatsa mai tsanani, tsada, da kuma yawan amfani da iskar oxygen a cikin ruwan sharar gida. Hanyar 2-isopropylnaphthalene da Kamfanin Cyanamide na Amurka ya ƙirƙiro tana amfani da naphthalene da propylene a matsayin kayan da aka samar don samar da 2-naphthol a matsayin wani abu da ya rage na acetone. Wannan hanyar tana kama da samar da phenol ta hanyar cumene. Adadin amfani da kayan: naphthalene mai tsafta 1170kg/t, sulfuric acid 1080kg/t, solid caustic soda 700kg/t.
2.1) Hanyar narkewar alkali mai sulfonated. Sanya naphthalene mai ladabi a cikin tukunyar sulfonation sannan a dafa (narkewa) zuwa 140°C. Ƙara sinadarin sulfuric mai ƙarfi sau 1.085 (rabo na molar) cikin mintuna 20, ƙara zafin jiki, sannan a ajiye shi a 160-164°C na tsawon awanni 2.5. Amsar tana ƙarewa lokacin da abun ciki na 2-naphthalenesulfonic acid ya kai 66% ko fiye kuma jimlar acidity shine 25%-27%. Ana zuba sinadarin sulfonate a cikin tukunyar hydrolysis a 140-150℃ na tsawon awanni 1. Sannan a cikin tukunya mai tsaka tsaki, a tace shi da maganin sodium hydrogen nitrite a 80-90°C har sai takardar gwajin ja ta Congo ba ta canza launin shuɗi ba. Yi amfani da tururi da iska don fitar da iskar SO2, a bar ta ta hura zuwa 30-40°C sannan a tace tsotsa, a wanke da ruwan gishiri 10%, sannan a tace tsotsa don samun sodium 2-naphthalenesulfonate. Sanya sodium hydroxide a cikin tukunya mai narkewar alkali, a dumama (narkewa) zuwa 290°C, sannan a ƙara sodium 2-naphthalenesulfonate cikin kimanin awanni 3 har sai adadin alkaline mai kyauta ya kai 5%-6%. Bayan haka, an ajiye shi a 320-330°C na tsawon awa 1. Ana narkar da alkaline melt a cikin ruwa sannan a wuce SO2 a 70-80°C har sai phenolphthalein ya zama ba shi da launi. A zuba ruwa a tafasa sannan a wanke, a cire sodium sulfite, sannan a bushe sannan a tace a rage matsin lamba don samun samfurin da aka gama. Jimlar yawan amfanin shine 73%-74%.
2) Hanyar 2-isopropylnaphthalene. Littafin Chemicalbook, wanda ke amfani da naphthalene da propylene a matsayin kayan masarufi, yana sake haifar da 2-naphthol a lokaci guda kamar yadda aka samo daga acetone.
3. Hanyar shiri ita ce a ƙara sinadarin sulfuric acid a cikin naphthalene mai narkewa a 140°C, a yi amfani da sinadarin sulfonation a 162~164°C, a yi amfani da sinadarin hydrolyze na sulfonate, a hura naphthalene mai kyauta, sannan a yi aiki da sodium sulfite don samar da gishirin sodium acid 2-naphthalenesulfonic. Gishirin sodium mai ƙarfi da sodium hydroxide ana narkar da su ta hanyar alkali a 285-320°C, sannan a ajiye su a 320-330°C na tsawon awa 1. Ana narkar da narkar da alkali a cikin sulfur dioxide don ƙara acidity don samun samfurin da ba a so, wanda aka wanke kuma aka bushe sannan a tace shi don samun samfurin.
4. Hanyar narkewar alkali ta Sulfonation. Sanya naphthalene mai ladabi a cikin tukunyar sulfonation sannan a dafa (narkewa) zuwa 140°C. Ƙara sinadarin sulfur mai ƙarfi sau 1.085 (rabo na molar) cikin mintuna 20, ƙara zafin jiki, sannan a ajiye shi a 160~164℃ na tsawon awanni 2.5. Amsar za ta ƙare lokacin da abun ciki na 2-naphthalenesulfonic acid ya kai kashi 66% ko fiye kuma jimlar acidity shine 25%-27%. Ana zuba sinadarin sulfonate a cikin tukunyar hydrolysis a 140~150℃ na tsawon awanni 1. Sannan a cikin tukunyar neutralization, a tace shi da maganin sodium bisulfite a 80~90℃ har sai takardar gwajin ja ta Congo ba ta canza launin shuɗi ba. Yi amfani da tururi da iska don fitar da iskar SO2, a bar ta ta hura zuwa 30-40°C sannan a tace tsotsa, a wanke da ruwan gishiri 10%, sannan a tace tsotsa don samun sodium 2-naphthalenesulfonate. Sanya sodium hydroxide a cikin tukunya mai narkewar alkali, a dumama (narkewa) zuwa 290°C, sannan a ƙara sodium 2-naphthalenesulfonate cikin kimanin awanni 3 har sai adadin alkali mai kyauta ya kai 5% zuwa 6%. Bayan haka, an ajiye shi a 320-330°C na tsawon awa 1. Ana narkar da alkaline melt a cikin ruwa sannan a wuce SO2 a 70 ~ 80℃ har sai phenolphthalein ya zama ba shi da launi. A zuba ruwa a tafasa a wanke, a cire sodium sulfite, sannan a bushe a tace a rage matsin lamba don samun samfurin da aka gama. Jimlar yawan amfanin ƙasa shine 73% zuwa 74%. Hanyar 2-isopropylnaphthalene tana amfani da naphthalene da propylene a matsayin kayan aiki don samar da 2-naphthol yayin da acetone ke cikin samfurin.
Kayan da aka samo asali daga sama:sodium hydroxide–>sulfuric acid–> nitric acid–>anhydrous sodium sulfite–>sulfur dioxide–>naphthalene–>Congo ja test paper–>sodium bisulfite solution–> 2-naphthalenesulfonic acid–>Phenolphthalein–> Sodium 2-naphthalenesulfonate–>Sodium caustic soda mai ƙarfi
Kayayyakin da ke ƙasa: 2-naphthylamine–>R-1,1′-bin-2-naphthol–>Pigment Ja 21–>2-naphthylamine-1-sulfonic acid–>naphthylamine–>S -1,1′-Bi-2-naphthol–>1-Amino-2-naphthol-4-sulfonic acid–>Acid Shuɗi 74–>2-hydroxy-3-naphthoic acid–>2- Fluoronaphthalene–>6-Methoxy-2-acetnaphthalene–>baƙin chrome T–>6-hydroxynaphthalene-2-boronic acid–>2-naphthol-3,6-disulfonic acid disodium- ->1-Naphthyldiazo-2-hydroxy-4-sulfonic acid gishirin ciki–>2-Amino-8-naphthol-6-sulfonic acid–>Lithol Scarlet–>Pigment Ja 53:1 Littafin Sinadarai –>Pigment Orange 5–>Ja Mai Launi 4–>Ja Mai Launi 3–>Ja Mai Launi 17–>Naproxen–>Jami'in Tanning na Sinadarai HV–>Baƙi Mai Tsaka-tsaki 2S-RL–>Matsakaici Baƙi BL–>2-Naphthaleneboronic acid–>2-Hydroxy-1-naphthoic acid–>Sodium 6-hydroxy-2-naphthalenesulfonate–>N-phenyl-2-naphthylamine–>Jami'in Tanning na Sinadarai PNC–>2-naphthylamine-3,6,8-trisulfonic acid–>bis[3-hydroxy-4-[(2-hydroxy-1-naphthyl)azo]-1-naphthalenesulfonate Disodium hydrogen chromate–>Sodium 3-hydroxy-4-[(2-hydroxynaphthalene)azo]-7-nitronaphthalene-1-sulfonate–>Jami'in Tanning na Sinadarai Mai Lamba 9
Lokacin Saƙo: Afrilu-20-2021