Yanayin da ake ciki a yanzu: masana'antar magunguna ta fi mayar da hankali kan haɗakar sinadarai, magunguna na halitta da magungunan gargajiya na kasar Sin, kuma samarwa tana da halaye na samfura iri-iri, hanyoyin da suka yi rikitarwa da kuma ma'aunin samarwa daban-daban.
Ruwan sharar da ake samarwa ta hanyar amfani da magunguna yana da halaye na yawan gurɓata muhalli, abubuwan da ke tattare da shi, rashin gurɓata muhalli da kuma yawan gubar halittu.
Haɗa sinadarai da kuma fermentation na samar da magunguna ruwan sharar gida shine babban abin da ke haifar da matsalar gurbata muhalli a masana'antar magunguna.
Ruwan sharar da aka haɗa da sinadarai babban gurɓataccen abu ne da ake fitarwa yayin samar da magunguna [2].
Ruwan sharar magunguna za a iya raba shi zuwa rukuni huɗu [3], wato ruwan sharar da kuma ruwan uwa a tsarin samarwa;
Ruwan da ya rage a cikin murmurewa ya haɗa da ruwan da ke narkewa, ruwan da ake buƙata, samfurin da aka samo, da sauransu.
Magudanar ruwa ta hanyar amfani da kayan aiki kamar ruwan sanyaya, da sauransu.
Kayan aiki da ruwan sharar ƙasa;
Najasar gida.
Fasaha don magance matsalar sharar gida ta magunguna
Ganin halayen ruwan sharar gida na matsakaici kamar yawan COD, yawan nitrogen, yawan phosphorus, yawan gishiri, zurfin chroma, hadaddun sinadarai da rashin kyawun lalacewa, hanyoyin magani da aka fi amfani da su sun haɗa da maganin sinadarai da tsarin maganin sinadarai [6].
Dangane da nau'ikan ingancin ruwan sharar gida daban-daban, za a yi amfani da jerin hanyoyi kamar haɗakar tsarin kimiyyar lissafi da tsarin halittu [7].
Hoton
1. Fasahar magani ta jiki da sinadarai
A halin yanzu, manyan hanyoyin magance matsalar ruwa mai guba ta hanyar amfani da sinadarai don samar da sharar gida sun haɗa da: hanyar flotation na iskar gas, hanyar coagulation sedimentation, hanyar adsorption, hanyar reverse osmosis, hanyar ƙonewa da kuma tsarin oxidation mai zurfi [8].
Bugu da ƙari, hanyoyin electrolysis da hazo na sinadarai, kamar su micro-electrolysis na FE-C da hanyoyin hazo na MAP don cire nitrogen da phosphorus, ana kuma amfani da su sosai wajen magance matsalar ruwan sharar gida na tsaka-tsaki na magunguna.
1.1 Hanyar haɗakar ruwa da kuma rage ruwa
Tsarin haɗakar ƙwayoyin halitta tsari ne wanda ake canza ƙwayoyin da aka dakatar da su da ƙwayoyin colloidal a cikin ruwa zuwa yanayin rashin kwanciyar hankali ta hanyar ƙara sinadarai sannan a haɗa su zuwa ƙwayoyin halitta ko ƙwayoyin halitta waɗanda suke da sauƙin rabawa.
A halin yanzu, wannan fasaha galibi ana amfani da ita ne a lokacin da ake yin magani kafin a fara aiki, a tsakiyar aiki, da kuma a fannin kula da ruwan sharar da ake sha daga magunguna [10].
Fasahar coagulation da sedimentation tana da fa'idodin fasahar da ta girma, kayan aiki masu sauƙi, aiki mai ɗorewa da kuma kulawa mai dacewa.
Duk da haka, za a sami adadi mai yawa na laka mai guba da ake samarwa a lokacin amfani da wannan fasaha, wanda zai haifar da ƙarancin pH na fitar da ruwa da kuma yawan gishirin da ke cikin ruwan sharar.
Bugu da ƙari, fasahar coagulation da sedimentation ba za ta iya kawar da gurɓatattun abubuwa da ke narkewa a cikin ruwan shara yadda ya kamata ba, kuma ba za ta iya kawar da gurɓatattun abubuwa masu guba da cutarwa gaba ɗaya a cikin ruwan shara ba.
1.2 Hanyar ruwan sama ta sinadarai
Hanyar hazo ta sinadarai hanya ce ta sinadarai don kawar da gurɓatattun abubuwa a cikin ruwan shara ta hanyar amsawar sinadarai tsakanin sinadarai masu narkewa da gurɓatattun abubuwa a cikin ruwan shara don samar da gishiri, hydroxides ko hadaddun abubuwa masu rikitarwa.
Ruwan sharar gida na matsakaici na magunguna galibi yana ɗauke da babban yawan sinadarin ammonia nitrogen, phosphate da sulfate ions, da sauransu. Ga irin wannan ruwan sharar, ana amfani da hanyar hazo ta sinadarai don magani na zahiri da na sinadarai don tabbatar da aikin da aka saba yi na tsarin maganin sinadarai na gaba.
A matsayin fasahar gargajiya ta sarrafa ruwa, ana amfani da ruwan sama mai guba don rage wa ruwan shara laushi.
Saboda amfani da kayan sinadarai masu tsafta a cikin tsarin samar da ruwan sharar gida na matsakaiciyar magunguna, ruwan sharar sau da yawa yana ɗauke da yawan sinadarin ammonia nitrogen da phosphorus da sauran gurɓatattun abubuwa, ta amfani da hanyar hazo ta sinadarai ta magnesium ammonium phosphate na iya kawar da gurɓatattun abubuwa guda biyu yadda ya kamata a lokaci guda, kuma ana iya sake yin amfani da hazo ta gishirin magnesium ammonium phosphate da aka samar.
Hanyar hazo ta sinadarin magnesium ammonium phosphate kuma ana kiranta da hanyar struvite.
A cikin tsarin samar da magunguna na tsaka-tsaki, ana amfani da babban adadin sulfuric acid a wasu bita, kuma pH na wannan ɓangaren na ruwan sharar gida na iya zama ƙasa. Domin inganta ƙimar pH na ruwan sharar gida da kuma cire wasu ions na sulfate a lokaci guda, ana amfani da hanyar ƙara CaO sau da yawa, wanda ake kira hanyar hazo ta sinadarai ta hanyar desulfurization na sauri.
1.3 shaƙar iska
Ka'idar kawar da gurɓatattun abubuwa a cikin ruwan shara ta hanyar amfani da hanyar shara tana nufin amfani da kayan daskararru masu ramuka don sharar wasu ko nau'ikan gurɓatattun abubuwa a cikin ruwan shara, ta yadda za a iya cire ko sake yin amfani da gurɓatattun abubuwa a cikin ruwan shara.
Abubuwan da ake amfani da su a yau da kullun sun haɗa da tokar ƙura, slag, carbon da aka kunna da kuma resin adsorption, waɗanda daga cikinsu ake amfani da carbon da aka kunna sosai.
1.4 iska mai iyo
Hanyar iyo ta iska tsari ne na sarrafa ruwan shara inda ake amfani da ƙananan kumfa masu yaɗuwa sosai a matsayin masu ɗaukar kaya don samar da mannewa ga gurɓatattun abubuwa a cikin ruwan shara. Saboda yawan ƙananan kumfa da ke manne wa gurɓatattun abubuwa bai kai na ruwa ba kuma yana tashi sama, ana iya samun rabuwar ruwa mai ƙarfi ko ruwa-ruwa.
Siffofin iyo a iska sun haɗa da iyo a iska da aka narkar, iyo a iska da aka yi da iska, iyo a iska da kuma iyo a iska da aka yi da sinadarai, da sauransu [18], daga cikinsu akwai iyo a iska da aka yi da sinadarai da suka dace da maganin ruwan shara mai yawan abubuwan da aka dakatar.
Hanyar amfani da iska tana da fa'idodin ƙarancin saka hannun jari, tsari mai sauƙi, kulawa mai dacewa da ƙarancin amfani da makamashi, amma ba za ta iya kawar da gurɓatattun abubuwa da ke narkewa a cikin ruwan sharar gida yadda ya kamata ba.
1.5 electrolysis
Tsarin lantarki shine amfani da rawar da ake takawa a halin yanzu, samar da jerin halayen sinadarai, canza gurɓatattun abubuwa masu cutarwa a cikin ruwan sharar gida kuma an cire su. Ka'idar amsawar tsarin electrolytic da ke faruwa a cikin maganin electrolyte shine ta hanyar kayan lantarki da amsawar electrode, samar da sabbin iskar oxygen da hydrogen [H] na muhalli, kuma gurɓatattun ruwan sharar gida na amsawar REDOX yana kawar da gurɓatattun abubuwa.
Hanyar electrolysis tana da inganci mai kyau da sauƙin aiki a cikin maganin sharar gida. A lokaci guda, hanyar electrolysis na iya cire abubuwan da ke da launi a cikin ruwan sharar gida yadda ya kamata kuma yana inganta lalatawar ruwan sharar gida yadda ya kamata.
Hoton
2. Fasaha mai zurfi ta iskar shaka
Fasahar hada iskar shaka mai ci gaba, a matsayin sabuwar fasahar sarrafa ruwa, tana da fa'idodi da yawa, kamar ingantaccen aikin lalata gurɓatattun abubuwa, ƙarin lalacewa da kuma iskar shaka masu gurɓatawa da kuma rashin gurɓataccen abu na biyu.
Fasahar hada iskar shaka mai zurfi, wacce aka fi sani da fasahar hada iskar shaka mai zurfi, fasaha ce ta magani ta zahiri da sinadarai wadda ke amfani da sinadarin oxidizer, haske, wutar lantarki, sauti, maganadisu da kuma mai kara kuzari don samar da sinadarai masu aiki sosai (kamar ·OH) don lalata gurɓatattun abubuwa masu guba na halitta.
A fannin maganin sharar gida na magunguna, fasahar hada iskar shara ta zamani ta zama abin da bincike da kulawa suka mayar da hankali a kai.
Fasahar hada iskar shaka mai zurfi ta ƙunshi sinadaran iskar shaka mai amfani da lantarki, sinadarai masu haɗa iskar shaka, ultrasonic oxidation, wet catalytic oxidation, photocatalytic oxidation, composite catalytic oxidation, supercritical water oxidation da kuma ci gaba da fasahar hada iskar shaka mai amfani da lantarki.
Hanyar hada sinadarai ta hanyar hada sinadarai ita ce amfani da sinadarai da kansu ko kuma a wasu yanayi masu karfi wajen hada sinadarai don hada sinadarai masu gurbata muhalli a cikin ruwan shara domin cimma manufar kawar da gurbatattun abubuwa, hanyoyin hada sinadarai, gami da hada sinadarai masu gurbata muhalli, hanyar hada sinadarai masu gurbata muhalli da kuma hanyar hada sinadarai masu gurbata muhalli.
2.1 Tsarin hada iskar shaka na Fenton
Hanyar oxidation ta Fenton wani nau'i ne na hanyar oxidation mai ci gaba wanda ake amfani da shi sosai a halin yanzu. Wannan hanyar tana amfani da gishirin ferric (Fe2+ ko Fe3+) a matsayin mai kara kuzari don samar da ·OH tare da iskar shaka mai ƙarfi a ƙarƙashin yanayin ƙara H2O2, wanda zai iya samun amsawar oxidation tare da gurɓatattun abubuwa na halitta ba tare da zaɓi ba don cimma lalata da kuma ƙara ma'adinai na gurɓatattun abubuwa.
Wannan hanyar tana da fa'idodi da yawa, gami da saurin amsawa da sauri, babu gurɓataccen abu na biyu da kuma ƙarfin iskar shara, da sauransu. Ana amfani da hanyar iskar shara ta Fenton a cikin maganin sharar gida na magunguna saboda rashin zaɓin amsawar iskar shara a cikin tsarin iskar shara na sinadarai kuma hanyar na iya rage gubar ruwan shara da sauran halaye.
2.2 Hanyar hada iskar shaka ta lantarki
Hanyar haɗakar iskar gas ta lantarki ita ce amfani da kayan lantarki don samar da radicals masu ɗauke da superoxide ·O2 da hydroxyl ·OH, waɗanda dukkansu suna da yawan aiki na haɗakar iskar gas, suna iya lalata kwayoyin halitta a cikin ruwan sharar gida, sannan su cimma manufar kawar da gurɓatattun abubuwa.
Duk da haka, wannan hanyar tana da halaye na yawan amfani da makamashi da kuma tsadar sa.
2.3 Iskar shaka ta Photocatalytic
Iskar sharar gida ta Photocatalytic wata fasaha ce ta magani mai inganci a fannin fasahar sarrafa ruwa, wadda ke amfani da kayan kariya (kamar TiO2, SrO2, WO3, SnO2, da sauransu) a matsayin masu ɗaukar sinadarai masu guba don aiwatar da iskar sharar gida ta yawancin gurɓatattun abubuwa da ke rage gurɓatattun abubuwa a cikin ruwan shara, don cimma manufar kawar da gurɓatattun abubuwa.
Saboda yawancin abubuwan da ke cikin ruwan sharar magunguna abubuwa ne na polar tare da ƙungiyoyin acidic ko abubuwa na polar tare da ƙungiyoyin alkaline, irin waɗannan abubuwa na iya lalacewa kai tsaye ko a kaikaice ta hanyar haske.
2.4 Iskar shaka mai ƙarfi ta ruwa
Oxidation na ruwa mai tsanani (SCWO) wani nau'in fasahar sarrafa ruwa ne wanda ke ɗaukar ruwa a matsayin matsakaici kuma yana amfani da halaye na musamman na ruwa a cikin yanayin supercritical don inganta ƙimar amsawa da kuma cimma cikakken oxidation na abubuwan halitta.
2.5 Fasaha mai haɗakar iskar shaka mai zurfi
Kowace fasahar hada iskar shara ta zamani tana amfani da nata iyakokin, domin inganta ingancin maganin sharar gida, ana hada jerin fasahar hada iskar shara ta zamani tare, wanda ya samar da hadewar fasahar hada iskar shara ta zamani, ko kuma wata fasahar hada iskar shara ta zamani tare da wasu fasahohi zuwa sabuwar fasaha don inganta karfin hada iskar shara da tasirin magani, da kuma biyan sauye-sauyen ingancin ruwa a manyan hanyoyin magance sharar gida na magunguna.
UV-Fenton, UV-H2O2, UV-O3, ultrasonic photocatalysis, kunna carbon photocatalysis, microwave photocatalysis da photocatalysis, da sauransu. A halin yanzu, fasahar haɗin ozone da aka fi nazari a kai sune [36]:
Tsarin carbon da aka kunna daga ozone, O3-H2O2 da UV-O3, sakamakon tasirin maganin ruwan sharar gida mai hana ruwa gudu da aikace-aikacen injiniya, O3-H2O2 da UV-O3 suna da ƙarin damar haɓakawa.
Tsarin haɗakar Fenton da aka saba amfani da shi ya haɗa da hanyar Fenton mai amfani da micro-electrolysis, hanyar ɗaukar ƙarfe ta H2O2, hanyar Fenton mai amfani da photochemical (kamar hanyar Fenton ta hasken rana, hanyar UV-Fenton, da sauransu), amma ana amfani da hanyar Fenton ta lantarki sosai.
Hoton
3. Fasahar maganin sinadarai ta Biochemical
Fasahar maganin sinadarai ta Biochemical ita ce babbar fasahar da ake amfani da ita wajen magance matsalar ruwa, ta hanyar girma, metabolism, haifuwa da sauran hanyoyin da za a bi wajen lalata kwayoyin halitta a cikin ruwan shara, samun makamashin da ake bukata da kuma cimma manufar kawar da kwayoyin halitta.
3.1 Fasahar maganin halittu ta anaerobic
Fasahar maganin halittu ta anaerobic ba ta da yanayin iskar oxygen na kwayoyin halitta, amfani da metabolism na kwayoyin cuta na anaerobic, ta hanyar tsarin hydrolytic acidification, samar da hydrogen acetic acid da samar da methane da sauran hanyoyin canza macromolecules, wanda ke da wahalar lalata kwayoyin halitta zuwa CH4, CO2, H2O da ƙananan kwayoyin halitta.
Ruwan sharar da aka yi da roba sau da yawa yana ƙunshe da adadi mai yawa na abubuwa masu rai waɗanda ba za a iya lalata su kai tsaye ta hanyar ƙwayoyin cuta masu amfani da iskar oxygen ba, don haka fasahar maganin anaerobic ta yanzu ta zama babbar hanyar magance sharar magunguna a gida da waje [43].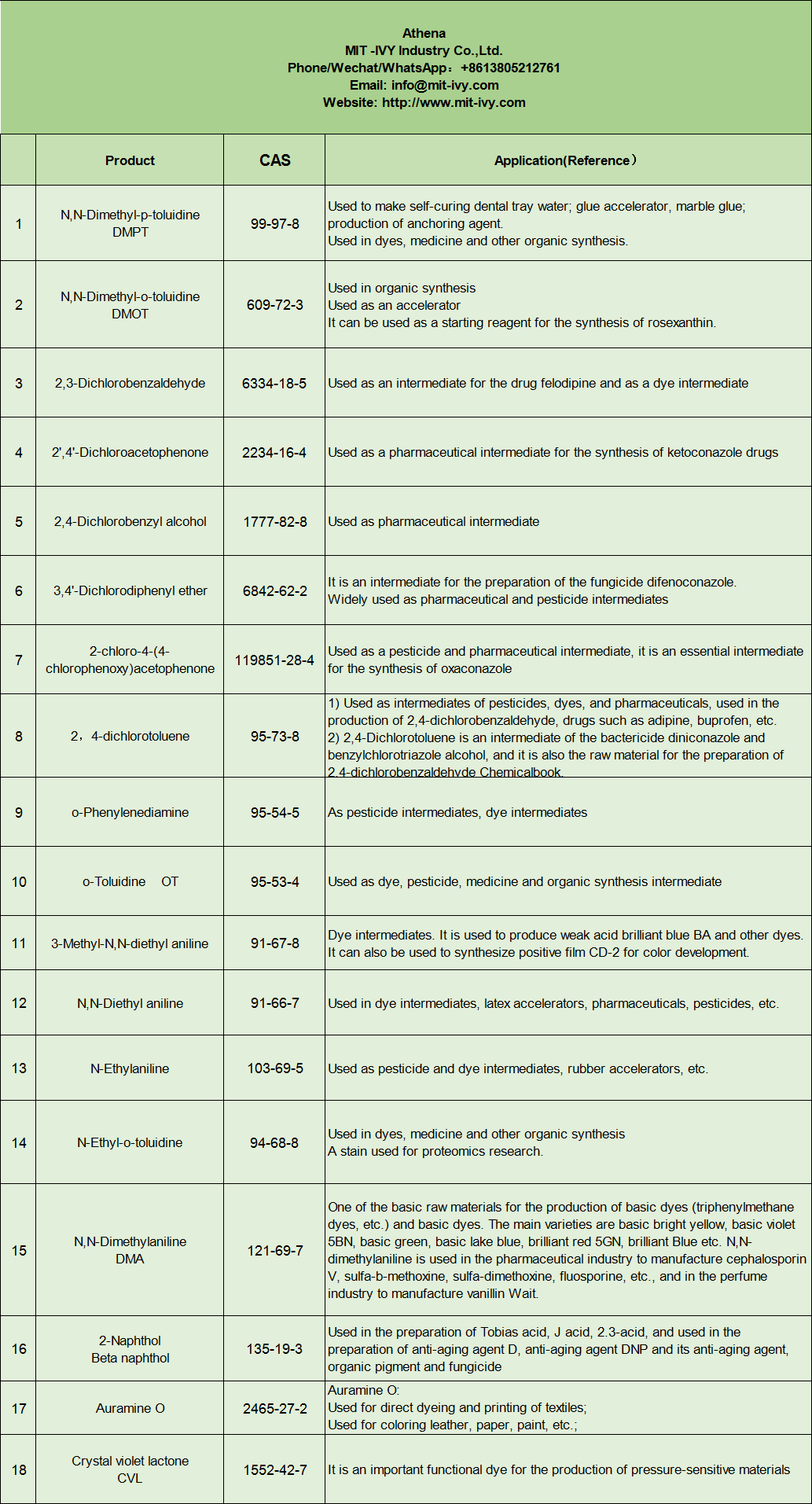
Fasahar maganin halittu ta anaerobic tana da fa'idodi da yawa: tsarin aiki na anaerobic reactor ba ya buƙatar samar da iska, yawan amfani da makamashi yana da ƙasa;
Yawan ruwa mai tasiri na anaerobic yana da yawa gabaɗaya.
Ƙananan buƙatun abinci mai gina jiki;
Yawan sinadarin anaerobic reactor yana da ƙasa, kuma sinadarin yana da sauƙin bushewa.
Ana iya sake amfani da methane da aka samar a cikin tsarin anaerobic azaman makamashi.
Duk da haka, ba za a iya fitar da ruwan da ke fitar da ruwa zuwa ga ƙa'ida ba, kuma yana buƙatar a ƙara magance shi ta hanyar haɗa shi da wasu hanyoyin. Duk da haka, fasahar maganin halittu ta anaerobic tana da alaƙa da ƙimar pH, zafin jiki da sauran abubuwa. Idan canjin ya yi yawa, amsawar anaerobic za ta shafi kai tsaye, sannan ingancin ruwan zai shafi.
3.2 Fasahar maganin halittu masu kama da iska
Fasahar maganin halittu ta Aerobic wata fasaha ce ta maganin halittu wadda ke amfani da rugujewar oxidative da kuma haɗa ƙwayoyin cuta masu guba don cire abubuwan da suka lalace. A lokacin girma da kuma metabolism na ƙwayoyin cuta masu guba, za a yi yawan haifuwa, wanda zai samar da sabon laka da aka kunna. Za a fitar da laka da aka kunna da ya wuce kima ta hanyar laka da ya rage, kuma za a tsarkake ruwan shara a lokaci guda.
| Samfuri | CAS |
| N,N-Dimethyl-p-toluidine DMPT | 99-97-8 |
| N,N-Dimethyl-o-toluidine DMOT | 609-72-3 |
| 2,3-Dichlorobenzaldehyde | 6334-18-5 |
| 2′,4′-Dichloroacetophenone | 2234-16-4 |
| Barasa na 2,4-Dichlorobenzyl | 1777-82-8 |
| 3,4′-Dichlorodiphenyl ether | 6842-62-2 |
| 2-chloro-4-(4-chlorophenoxy)acetophenone | 119851-28-4 |
| 2, 4-dichlorotoluene | 95-73-8 |
| o-Phenylenediamine | 95-54-5 |
| o-Toluidine OT | 95-53-4 |
| 3-Methyl-N,N-diethyl aniline | 91-67-8 |
| N,N-Diethyl aniline | 91-66-7 |
| N-Ethylaniline | 103-69-5 |
| N-Ethyl-o-toluidine | 94-68-8 |
| N,N-Dimethylaniline DMA | 121-69-7 |
| 2-Naphthol Beta naphthol | 135-19-3 |
| Auramin O | 2465-27-2 |
| Lactone mai launin shuɗi mai launin crystal CVL | 1552-42-7 |
Masana'antar Sinadarai ta MIT –IVY tare daMasana'antu 4na tsawon shekaru 19, riniMatsakaicis & tsaka-tsakin magunguna &sinadarai masu kyau da na musamman .TEL(WhatsApp):008613805212761 Athena
Lokacin Saƙo: Afrilu-25-2021